- Details
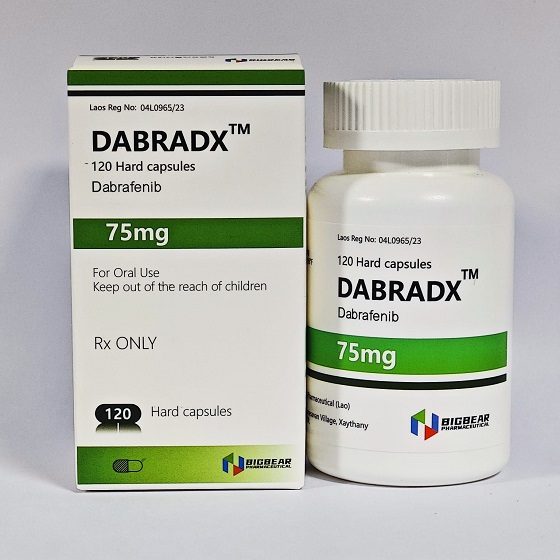
- Description
-
Packaging Size120C/bottle
-
Strength75mg
-
CompositonDabrafenib
-
TreatmentBRAF V600E Mutation-Positive Solid Tumors,Melanoma,Non-Small Cell Lung Cancer,Locally Advanced or Metastatic Thyroid Cancer,
-
FormCapsule
-
BrandDABRADX
-
Quantity Unit75mg*120C/bottle
-
ManufacturerBIGBEAR Pharma,Laos PDR
Dabrafenib is an anti-cancer medication used for the treatment of cancers associated with a mutated version of the gene BRAF.Dabrafenib acts as an inhibitor of the associated enzyme B-Raf, which plays a role in the regulation of cell growth.
Melanoma
BRAF V600E mutation-positive unresectable or metastatic melanoma
- Indicated as a single agent
- 150 mg PO BID
- Continue until disease recurrence or unacceptable toxicity
BRAF V600E or V600K mutation-positive unresectable or metastatic melanoma
- Indicated, in combination with trametinib
- 150 mg PO BID plus trametinib 2 mg PO qDay
- Continue until disease recurrence or unacceptable toxicity
Adjuvant treatment of BRAF V600E or V600K mutation-positive melanoma
- Indicated in combination with trametinib when lymph node(s) involved following complete resection
- 150 mg PO BID plus trametinib 2 mg PO qDay
- Continue until disease recurrence or unacceptable toxicity for up to 1 year
Non-Small Cell Lung Cancer
BRAF V600E mutation-positive
Indicated in combination with trametinib
150 mg PO BID plus trametinib 2 mg PO qDay
Continue until disease recurrence or unacceptable toxicity
Also see Administration
Thyroid Cancer, Locally Advanced or Metastatic
BRAF V600E mutation-positive
Indicated, in combination with trametinib, for locally advanced or metastatic anaplastic thyroid cancer (ATC) in adults with no satisfactory locoregional treatment options
150 mg PO BID plus trametinib 2 mg PO qDay
Continue until disease recurrence or unacceptable toxicity
BRAF V600E Mutation-Positive Solid Tumors
Indicated in combination with trametinib for unresectable or metastatic solid tumors with BRAF V600E mutation in patients who have progressed following prior treatment and have no satisfactory alternative treatment options
150 mg PO BID plus trametinib 2 mg PO qDay
Continue until disease recurrence or unacceptable toxicity