- Details
- Description
-
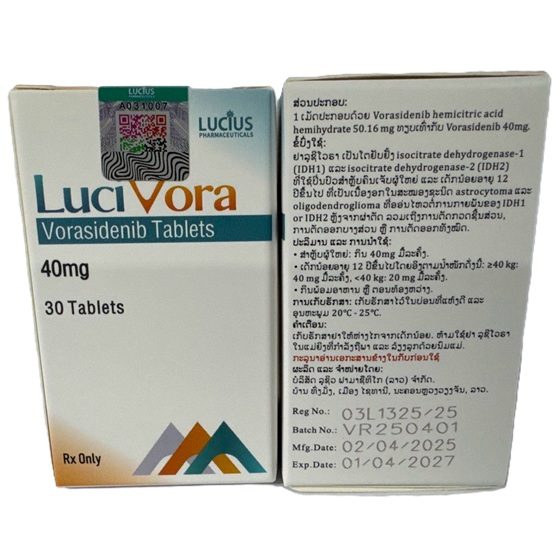
Packaging Size30t/Bottle
-
Strength40mg
-
CompositonVorasidenib
-
TreatmentGrade 2 IDH-mutant astrocytoma or oligodendroglioma
-
FormTablet
-
BrandLuciVora
-
Quantity Unit40mg*30t/Box
-
ManufacturerLucius Pharmaceuticals (Lao) Co.,Ltd
About Vorasidenib
Vorasidenib is an oral, brain-penetrant, dual inhibitor of mutant isocitrate dehydrogenase 1 (IDH1) and 2 (IDH2) enzymes, approved for treating grade 2 IDH-mutant astrocytoma or oligodendroglioma in adults and children 12 years and older, following surgery.
Adult
Astrocytoma or Oligodendroglioma
Indicated for Grade 2 astrocytoma or oligodendroglioma with susceptible isocitrate dehydrogenase (IDH)-1 or IDH-2 mutation following surgery including biopsy, sub-total resection, or gross total resection
40 mg PO qDay until disease progression or unacceptable toxicity
Dosage Modifications
Recommended dosage reductions
- First reduction: 20 mg PO qDay
- Second reduction: 10 mg PO qDay
- Permanently discontinue if second dosage reduction not tolerated
Hepatotoxicity
-
ALT or AST >ULN to 3 x ULN without concurrent total bilirubin >2 x ULN
- Continue current dose
- Monitor liver function tests (LFTs) weekly until recovery to
-
ALT or AST >3-5 x ULN without concurrent total bilirubin >2 x ULN
- Hold until recovery to ≤Grade 1 or baseline
- Resume at same dose for recovery ≤28 days or at reduced dose for recovery >28 days
- Recurrence: Hold therapy until recovery to ≤Grade 1 or baseline and resume at reduced dose
-
ALT or AST >5-20 x ULN without concurrent total bilirubin >2 x ULN
- Hold therapy until recovery to ≤Grade 1 or baseline
- Resume at reduced dose for recovery ≤28 days or permanently discontinue for recovery >28 days
- Recurrence: Permanently discontinue
-
ALT or AST >3-20 x ULN with concurrent total bilirubin >2 x ULN
- Hold therapy until recovery to ≤Grade 1 or baseline
- Resume at reduced dose
- Recurrence: Permanently discontinue
-
Any ALT or AST >20 x ULN
- Permanently discontinue
Other adverse reactions
-
Grade 3
- Hold therapy until recovery to ≤Grade 1 or baseline
- Resume at reduced dose
- Recurrence: Permanently discontinue
-
Grade 4
- Permanently discontinue
Renal impairment
- CrCl >40 mL/min: No dosage adjustment necessary
- CrCl ≤40 mL/min or on dialysis: Not studied; monitor for adverse reactions and adjust dose as recommended
Hepatic impairment
- Mild or moderate (Child-Pugh class A or B): No dosage adjustment necessary
- Severe (Child-Pugh class C): Not studied; monitor for adverse reactions and adjust dose as recommended
Dosing Considerations
Evaluate blood chemistry and LFTs before initiating
Patient selection
- Test for presence of IDH1 or IDH2 mutations in tumor specimens
Pediatric
Astrocytoma or Oligodendroglioma
Indicated for Grade 2 astrocytoma or oligodendroglioma with susceptible isocitrate dehydrogenase (IDH)-1 or IDH-2 mutation following surgery including biopsy, sub-total resection, or gross total resection in patients ≥12 years
≥40 kg: 40 mg PO qDay
<40 kg: 20 mg PO qDay
Continue until disease progression or unacceptable toxicity
Safety and efficacy not established in patients <12 years
Dosage Modifications
Recommended dosage adjustments
-
≥40 kg
- First reduction: 20 mg PO qDay
- Second reduction: 10 mg PO qDay
-
<40 kg
- First reduction: 10 mg PO qDay
- Permanently discontinue if reduced dose not tolerated
Hepatotoxicity
-
ALT or AST >ULN to 3 x ULN without concurrent total bilirubin >2 x ULN
- Continue current dose
- Monitor liver function tests (LFTs) weekly until recovery to
-
ALT or AST >3-5 x ULN without concurrent total bilirubin >2 x ULN
- Hold until recovery to ≤Grade 1 or baseline
- Resume at same dose for recovery ≤28 days or at reduced dose for recovery >28 days
- Recurrence: Hold therapy until recovery to ≤Grade 1 or baseline and resume at reduced dose
-
ALT or AST >5-20 x ULN without concurrent total bilirubin >2 x ULN
- Hold therapy until recovery to ≤Grade 1 or baseline
- Resume at reduced dose for recovery ≤28 days or permanently discontinue for recovery >28 days
- Recurrence: Permanently discontinue
-
ALT or AST >3-20 x ULN with concurrent total bilirubin >2 x ULN
- Hold therapy until recovery to ≤Grade 1 or baseline
- Resume at reduced dose
- Recurrence: Permanently discontinue
-
Any ALT or AST >20 x ULN
- Permanently discontinue
Other adverse reactions
-
Grade 3
- Hold therapy until recovery to ≤Grade 1 or baseline
- Resume at reduced dose
- Recurrence: Permanently discontinue
-
Grade 4
- Permanently discontinue
Renal impairment
- CrCl >40 mL/min: No dosage adjustment necessary
- CrCl ≤40 mL/min or on dialysis: Not studied; monitor for adverse reactions and adjust dose as recommended
Hepatic impairment
- Mild or moderate (Child-Pugh class A or B): No dosage adjustment necessary
- Severe (Child-Pugh class C): Not studied; monitor for adverse reactions and adjust dose as recommended
Dosing Considerations
Evaluate blood chemistry and LFTs before initiating
Patient selection
- Test for presence of IDH1 or IDH2 mutations in tumor specimens