- Details
- Description
-
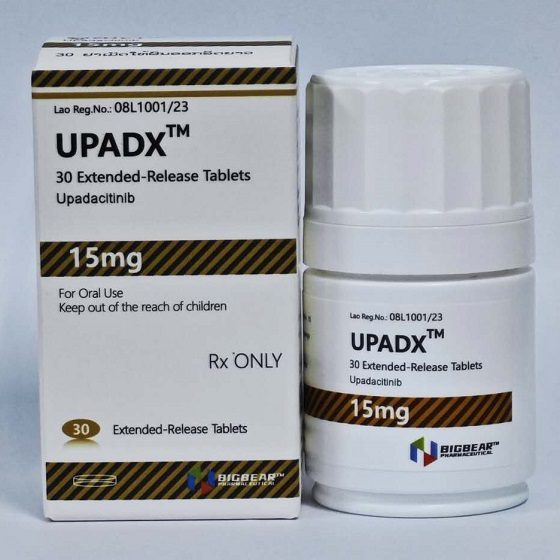
Packaging Size30T/bottle
-
Strength15mg
-
CompositonUpadacitinib
-
TreatmentRheumatoid Arthritis, Psoriatic Arthritis, Atopic Dermatitis,Ulcerative Colitis, Crohn's Disease, Ankylosing Spondylitis, and Axial Spondyloarthritis.
-
FormTablet
-
BrandUPADX
-
Quantity Unit15mg*30T/bottle
-
ManufacturerBIGBEAR Pharma,Laos PDR
Upadacitinib is a medication used for the treatment of rheumatoid arthritis, psoriatic arthritis, atopic dermatitis, ulcerative colitis, Crohn's disease, ankylosing spondylitis, and axial spondyloarthritis.Upadacitinib is a Janus kinase (JAK) inhibitor that works by blocking the action of enzymes called Janus kinases. These enzymes are involved in setting up processes that lead to inflammation, and blocking their effect brings inflammation in the joints under control.
Common side effects include upper respiratory tract infections (common cold, sinus infections), nausea, cough, and fever.
Rheumatoid Arthritis
Indicated for moderately to severely active rheumatoid arthritis (RA) in adults who have had an inadequate response or intolerance to ≥1 tumor necrosis factor (TNF) blockers
15 mg PO qDay
Psoriatic Arthritis
Indicated for active psoriatic arthritis (PsA) in adults who have had an inadequate response or intolerance to ≥1 TNF blockers
15 mg PO qDay
Atopic Dermatitis
Indicated for refractory moderate-to-severe atopic dermatitis (AD) in adults whose disease is not adequately controlled with other systemic therapies or if those therapies are inadvisable
<65 years
- 15 mg PO qDay initially; consider increasing to 30 mg qDay if an adequate response is not achieved
- Discontinue if 30-mg dose if adequate response not achieved
- Use lowest effective dose needed to maintain response
Ulcerative Colitis
Indicated for moderate to severe active ulcerative colitis (UC) in adults who had inadequate response or intolerance to ≥1 TNF blocker
Induction: 45 mg PO qDay x 8 weeks
Maintenance
- 15 mg PO qDay
- Refractory, severe, or extensive disease: Consider 30 mg qDay
- Use lowest effective dosage needed to maintain response
- Discontinue if unable to achieve adequate therapeutic response with 30 mg/day
Ankylosing Spondylitis
Indicated for active ankylosing spondylitis (AS) in adults who have had an inadequate response or intolerance to ≥1 TNF blockers
15 mg PO qDay
Nonradiographic Axial Spondyloarthritis
Indicated for active nonradiographic axial spondyloarthritis (nr-axSpA) with objective signs of inflammation who have had an inadequate response or intolerance to TNF blocker therapy
15 mg PO qDay
Crohn Disease
Indicated for moderately-to-severely active Crohn disease in adults who have had an inadequate response or intolerance to ≥1 TNF blockers
Induction: 45 mg PO qDay x 12 weeks
Maintenance: 15 mg PO qDay; consider 30 mg qDay for patients with refractory, severe, or extensive disease
Discontinue if unable to achieve an adequate therapeutic response with 30-mg dosage
Use lowest effective dose needed to maintain response
Dosage Modifications
Coadministration of strong CYP3A4 inhibitors
- RA, PsA, AD, AS, or nr-axSpA: 15 mg PO qDay
- UC: 30 mg qDay x 8 weeks for induction, then 15 mg qDay for maintenance
- CD: 30 mg qDay x 12 weeks for induction, then 15 mg qDay for maintenance
Infection
- If serious infection (eg, serious opportunistic infections) develops, interrupt treatment until infection is controlled
Dose interruption for laboratory abnormalities
- Absolute neutrophil count (ANC) <1000 cells/mm3: Interrupt; may restart once ANC >1000 cells/mm3
- Absolute lymphocyte count (ALC) <500 cells/mm3: Interrupt; may restart once ALC >500 cells/mm3
- Hemoglobin (Hb) <8 g/dL: Interrupt; may restart once Hb >8 g/dL
- Hepatic transaminases: Interrupt if drug-induced liver injury suspected
Renal impairment
-
RA, PsA, AS, or nr-axSpA
- Mild (eGFR 60 to < 90 mL/min/1.73 m2), moderate (eGFR 30 to < 60 mL/min/1.73 m2), severe renal impairment (eGFR 15 to < 30 mL/min/1.73 m2): No dose adjustment necessary
- ESRD: Not studied
-
AD
- Mild or moderate (eGFR >30 mL/min/1.73 m2): No dose adjustment necessary
- Severe (eGFR <30 mL/min/1.73 m2): 15 mg PO qDay
- ESRD: Not studied
-
UC
- Mild or moderate (eGFR >30 mL/min/1.73 m2): No dose adjustment necessary
- Severe (eGFR 15 to <30 mL/min/1.73 m2): 30 mg qDay x 8 weeks for induction, then 15 mg qDay for maintenance
- ESRD (eGFR <15 mL/min/1.73 m2): Not recommended
-
Crohn disease
- Mild-to-moderate (eGFR ≥30 mL/min/1.73m2): No dosage adjustment necessary
- Severe (eGFR 15 to <30 mL/min/1.73m2): 30 mg qDay for 12 weeks for induction; 15 mg qDay for maintenance
- ESRD (eGFR ≥30 mL/min/1.73m2): Not recommended
Hepatic impairment
-
RA, PsA, AS, nr-axSpA, or AD
- Mild or moderate (Child-Pugh A or B): No dose adjustment required
-
RA, PsA, AS, nr-axSpA, UC, CD, or AD
-
Severe (Child-Pugh C): Not recommended
-
-
UC
- Mild or moderate (Child-Pugh A or B): 30 mg qDay x 8 weeks for induction, then 15 mg qDay for maintenance
- Severe (Child-Pugh C): Not recommended
-
Crohn disease
- Mild-to-moderate (Child-Pugh A or B): 30 mg qDay for 12 weeks for induction; 15 mg qDay for maintenance
- Severe (Child Pugh C): Not recommended
Dosing Considerations
ALC <500 cells/mm3, ANC <1000 cell/mm3, or Hb <8 g/dL: Do not initiate
-
Before initiating
- Test for tuberculosis (TB) before initiating; initiate antitubercular treatment for previously untreated latent TB or active TB before initiating treatment
- Verify pregnancy status in females of reproductive potential before initiating
- Screen viral hepatitis in accordance with clinical guidelines
- Update immunizations according to current immunization guidelines
-
Limitations of use
- RA, PsA, AS or nr-axSpA: Use in combination with other JAK inhibitors, biologic disease-modifying antirheumatic drugs (DMARDs), or with potent immunosuppressants (eg, azathioprine, cyclosporine), is not recommended
- AD: Not recommended in combination with other JAK inhibitors, biologic immunomodulators, or with other immunosuppressants
- UC, CD: Not recommended in combination with other JAK inhibitors, biological therapies, or potent immunosuppressants (eg, azathioprine, cyclosporine)