- Details
- Description
-
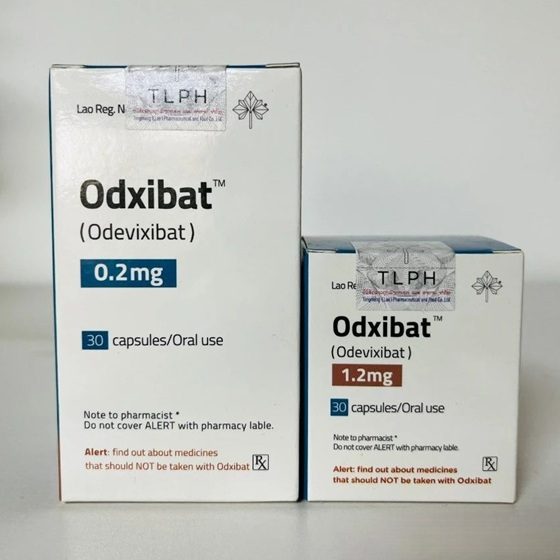
Packaging Size30c/bottle
-
Strength0.2mg&1.2mg
-
CompositonOdevixibat
-
TreatmentProgressive familial intrahepatic cholestasis
-
Formcapsules
-
BrandOdxibat
-
Quantity Unit0.2mg*30c/box&1.2mg*30c/box
-
ManufacturerTongmeng (Lao) Pharmaceutical & Food Co., Ltd.(TLPH)
About Odevixibat
Odevixibat is a medication for the treatment of progressive familial intrahepatic cholestasis. Odevixibat is a reversible, potent, selective inhibitor of the ileal bile acid transporter (IBAT).
Cholestasis-Induced Pruritus
Indicated for pruritus associated with progressive familial intrahepatic cholestasis (PFIC)
40 mcg/kg PO qAM
Weight-based dosing
- 45.5-55.4 kg: 2000 mcg PO qAM
- ≥55.5 kg: 2400 mcg PO qAM
- If no improvement after 3 months, may increase dose in 40-mcg/kg increments up to 120 mcg/kg/day; not to exceed 6 mg/day
Alagille Syndrome
Indicated for pruritus associated with Alagille syndrome (ALGS)
120 mcg/kg PO qAM
Weight-based dosing
- 45.5-55.4 kg: 6000 mcg PO qAM
- ≥55.5 kg: 7200 mcg PO qAM
Dosage Modifications
Tolerability for ALGS
- Consider dose reduction to 40 mcg/kg/day if tolerability issues occur in absence of other causes
- Increase to 120 mcg/kg/day once tolerability issues stabilize
Hepatic impairment
- Patients with PFIC may have impaired hepatic function at baseline
- Patients with clinically significant portal hypertension or patients with decompensated cirrhosis: Safety and efficacy not established
Liver abnormalities
- New onset of elevated liver function tests (LFTs) or symptoms consistent with clinical hepatitis: Interrupt therapy; once LFTs either return to baseline or stabilize at a new baseline value, consider restarting at recommended dose, and increase as tolerated if appropriate
- Elevated LFTs recur: Consider discontinuing permanently
- Hepatic decompensation event (eg, variceal hemorrhage, ascites, hepatic encephalopathy) occurs: Permanently discontinue
Dosing Considerations
Limitation of use
- Not effective in PFIC type 2 patients with ABCB11 variants resulting in nonfunctional or complete absence of bile salt export pump protein
Monitoring parameters
- Liver function tests (eg, ALT, AST, total bilirubin [TB], and direct bilirubin [DB]) and serum fat-soluble vitamins (FSV) levels: Baseline and during treatment
- International Normalized Ratio (INR): During treatment